 How to mix science with lollipops to appeal to Guides! This came from a leader training I attended several years ago.
How to mix science with lollipops to appeal to Guides! This came from a leader training I attended several years ago.
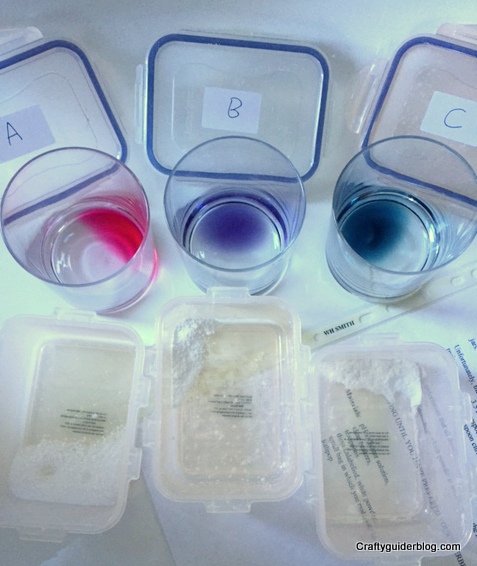
To prepare, you need to boil a red cabbage in water and leave it to stand for 30 minutes. Then drain the liquid into a container to use as indicator solution i.e. something that will distinguish between acids and alkalines.
equipment: 3 clean glasses/jars, teaspoons, small plastic bags (one per person), indicator solution (cabbage water), 3 containers
ingredients: icing sugar, bicarbonate of soda, citric acid (available from your chemist), lollipops
The experiment relies on the fact that citric acid is an acid and turns the indicator solution red, the bicarb is alkaline and turns it blue and the icing sugar is neutral and leaves it unchanged. You don’t need much powder and indicator solution to do this experiment, and please make sure the Guides don’t drink the cabbage water – I suppose it’s edible but it would taste foul!
I found that the older Guides had some concept of this type of experiment from school, however the younger ones hadn’t covered it yet. It might require a bit of leader/young leader supervision.
The printable sheet to hand to the Guides is on the link below.
I have also done this experiment with my Brownie and Rainbow daughters. They didn’t quite grasp the concept of acids and alkalines, but did enjoy the way the different substances changed the colour of the solution and asked questions along the way such as what would happen if all three substances were added at the same time.
